Effect of Temperature on Partial Miscibility

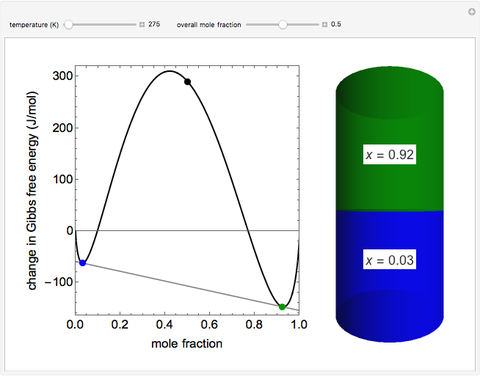
This simulation plots the change in Gibbs free energy versus mole fraction of one component in a non-ideal binary mixture. Two liquid phases form when the Gibbs free energy is lower for two phases than for one phase. A line that is tangent to the Gibbs free energy curve at two points (blue and green points) represents the Gibbs free energy change over the mole fraction range between the two points. The mole fractions at the blue and green points are the mole fractions of the two phases, and these mole fractions are shown on the container on the right. The size of each phase on the right is proportional to the amount of that phase, and these amounts are obtained from a mass balance (lever rule). Increasing the temperature with the slider decreases the range of partial miscibility. The slider on the right changes the overall mole fraction, and the black circle represents that mole fraction.
This simulation runs on desktop using the free Wolfram Player. Download the Wolfram Player here.
About:
Author: Kaiyuan Tang. Open content licensed under CC BY-NC-SA.
Additional contributions by: Rachael L. Baumann and John L. Falconer
View the source code for this simulation
