DePriester Chart for Hydrocarbons

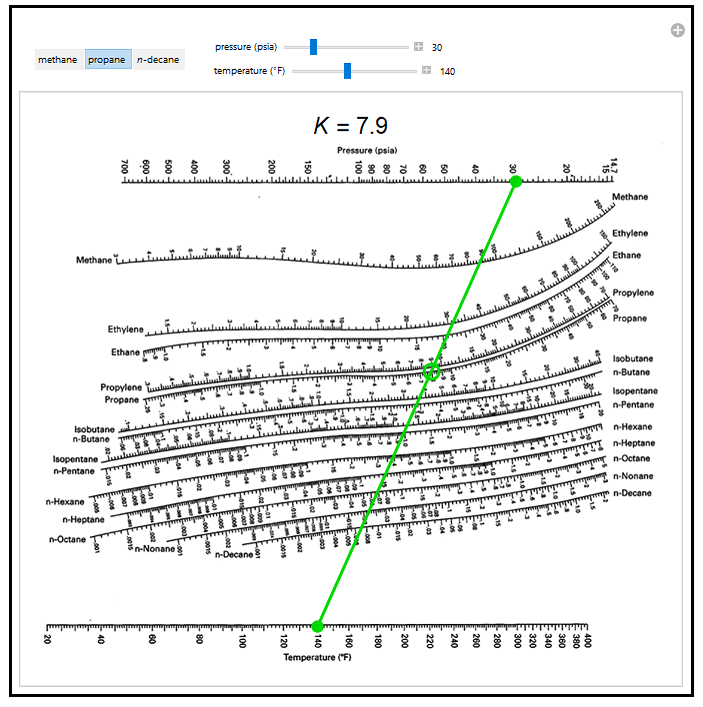
This simulation applies a DePriester chart, a set of nomograms, to find the vapor-liquid equilibrium ratio (the gas phase mole fraction divided by the liquid phase mole fraction, known as the K-value) for a series of hydrocarbons. Select one of three hydrocarbons: methane, propane, or n-decane. Use sliders to change the pressure and temperature; a green line connects these two points. The green circle indicates where to read the K-value on the chart, which is also displayed above the chart. The K-values for other hydrocarbons can also be read from the chart.
This simulation runs on desktop using the free Wolfram Player. Download the Wolfram Player here.
About:
This simulation was made at the University of Colorado Boulder, Department of Chemical and Biological Engineering. Author: Majed N. Aldossary
View the source code for this simulation
