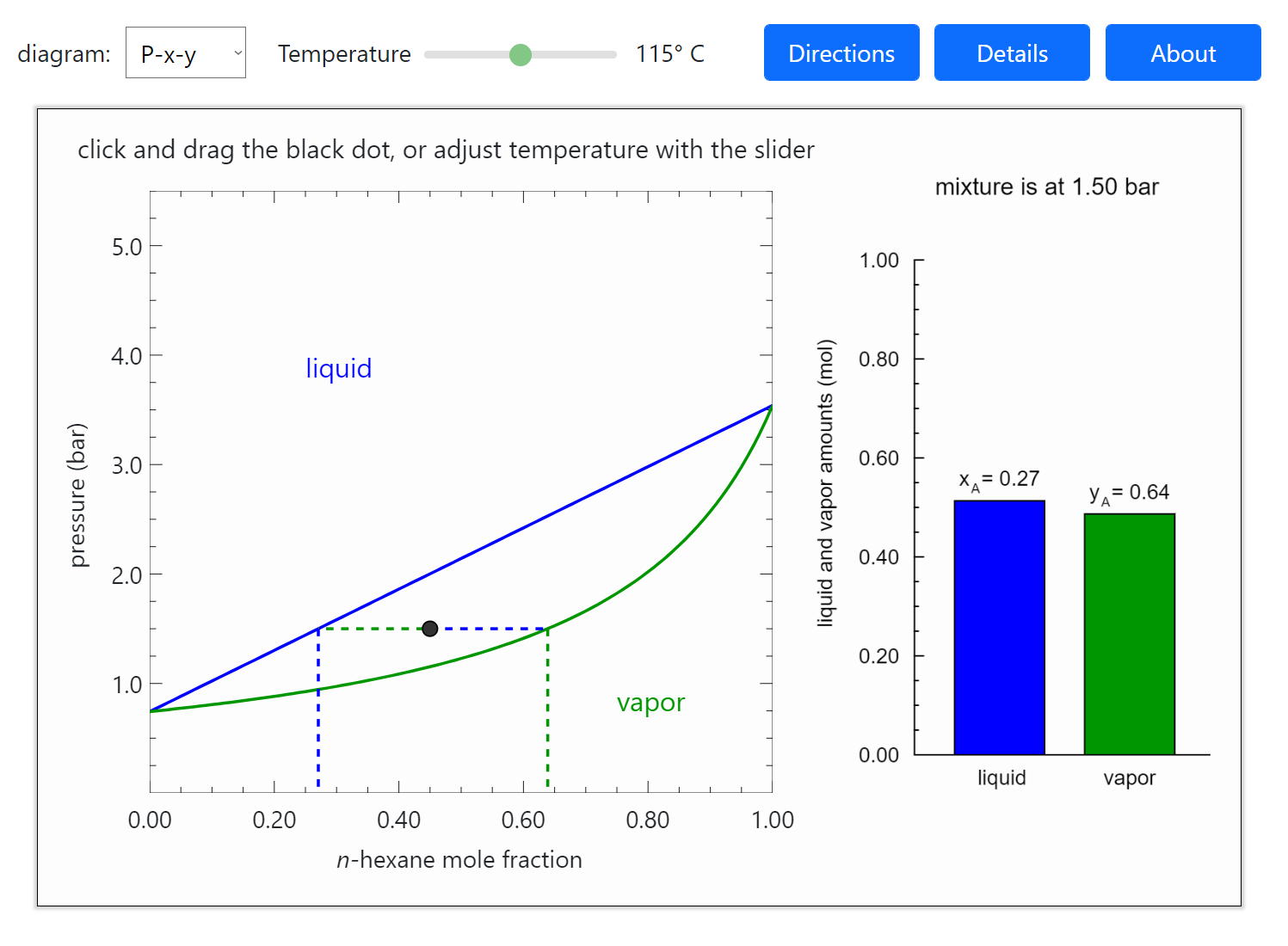
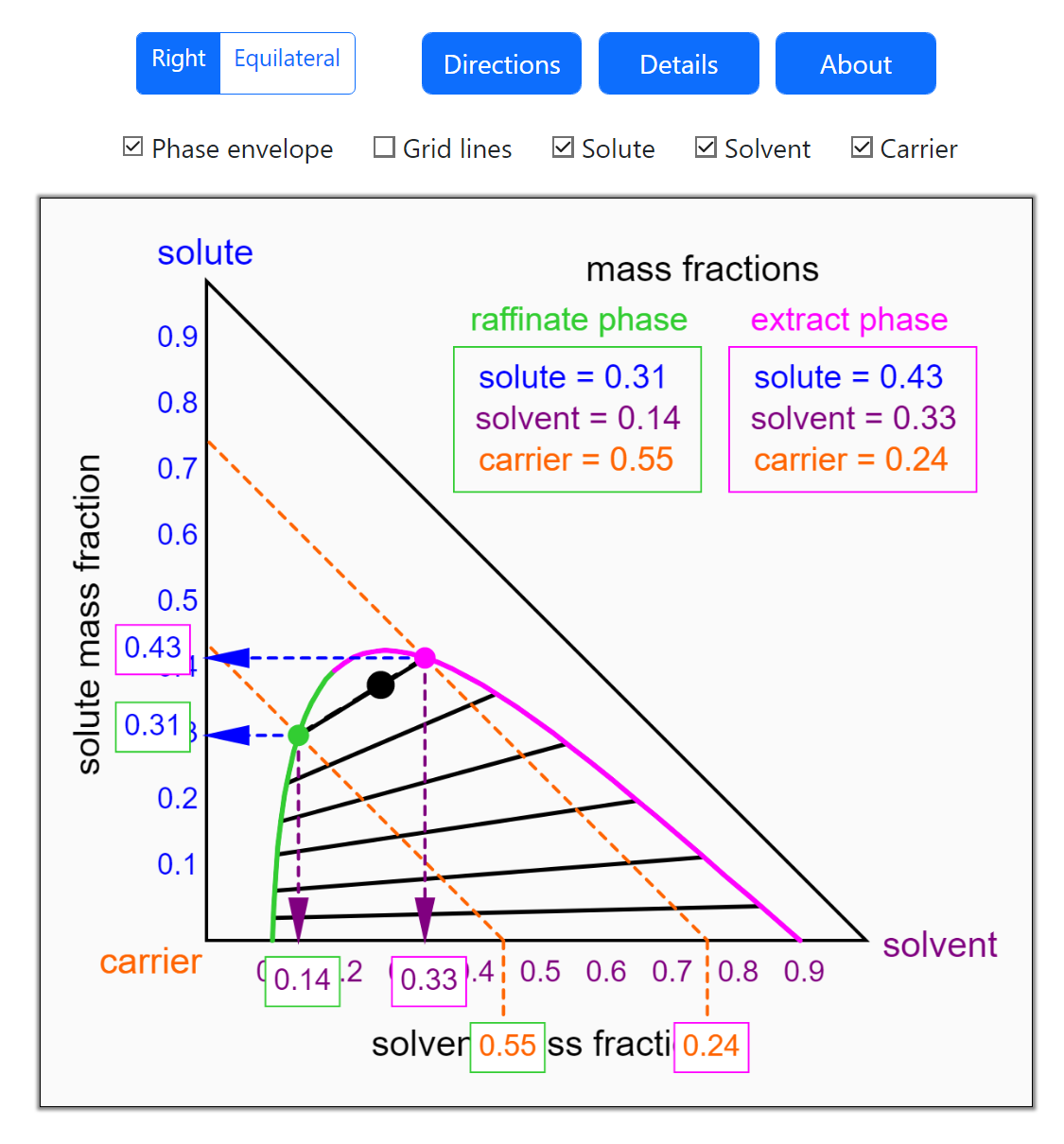
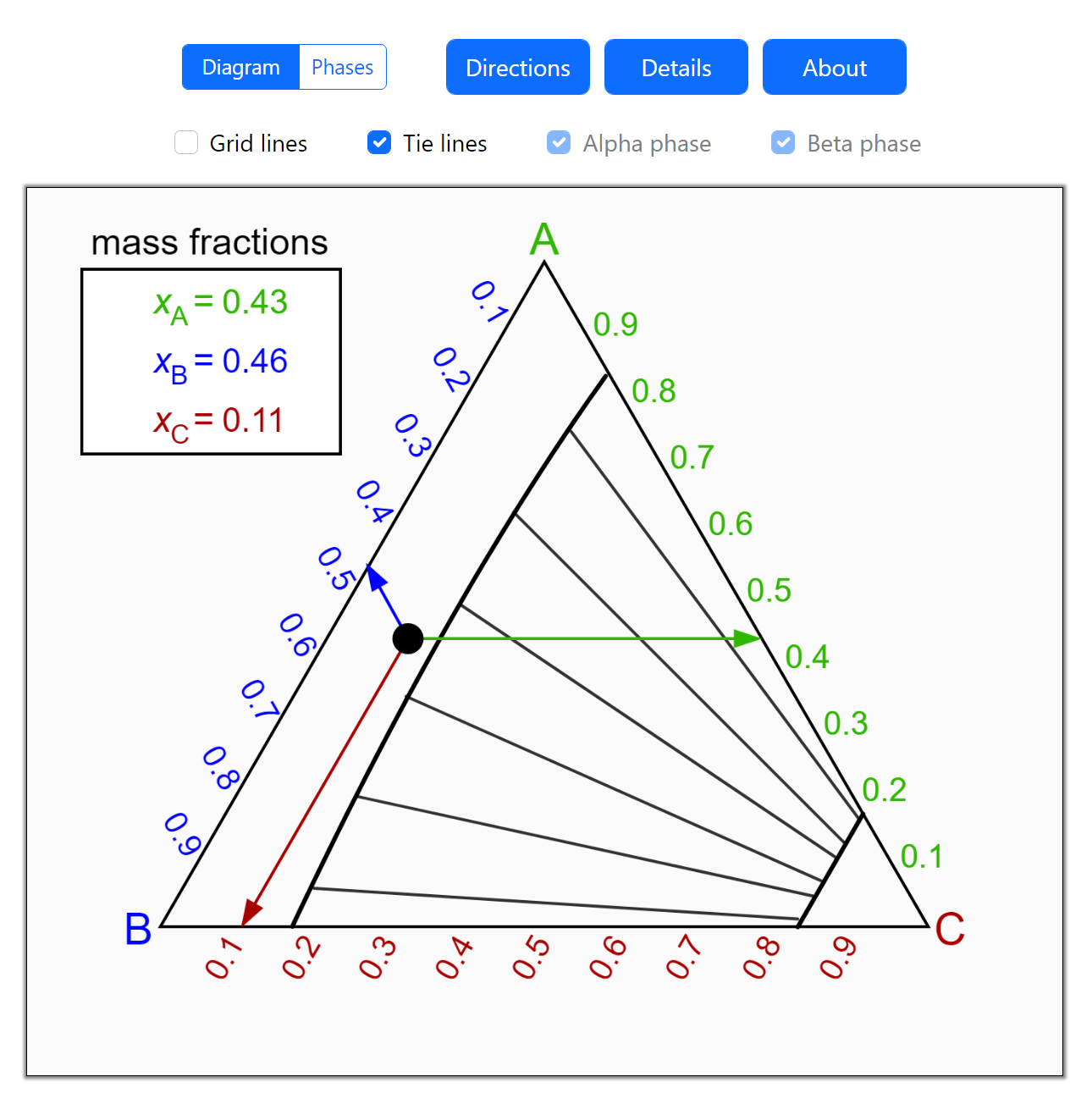
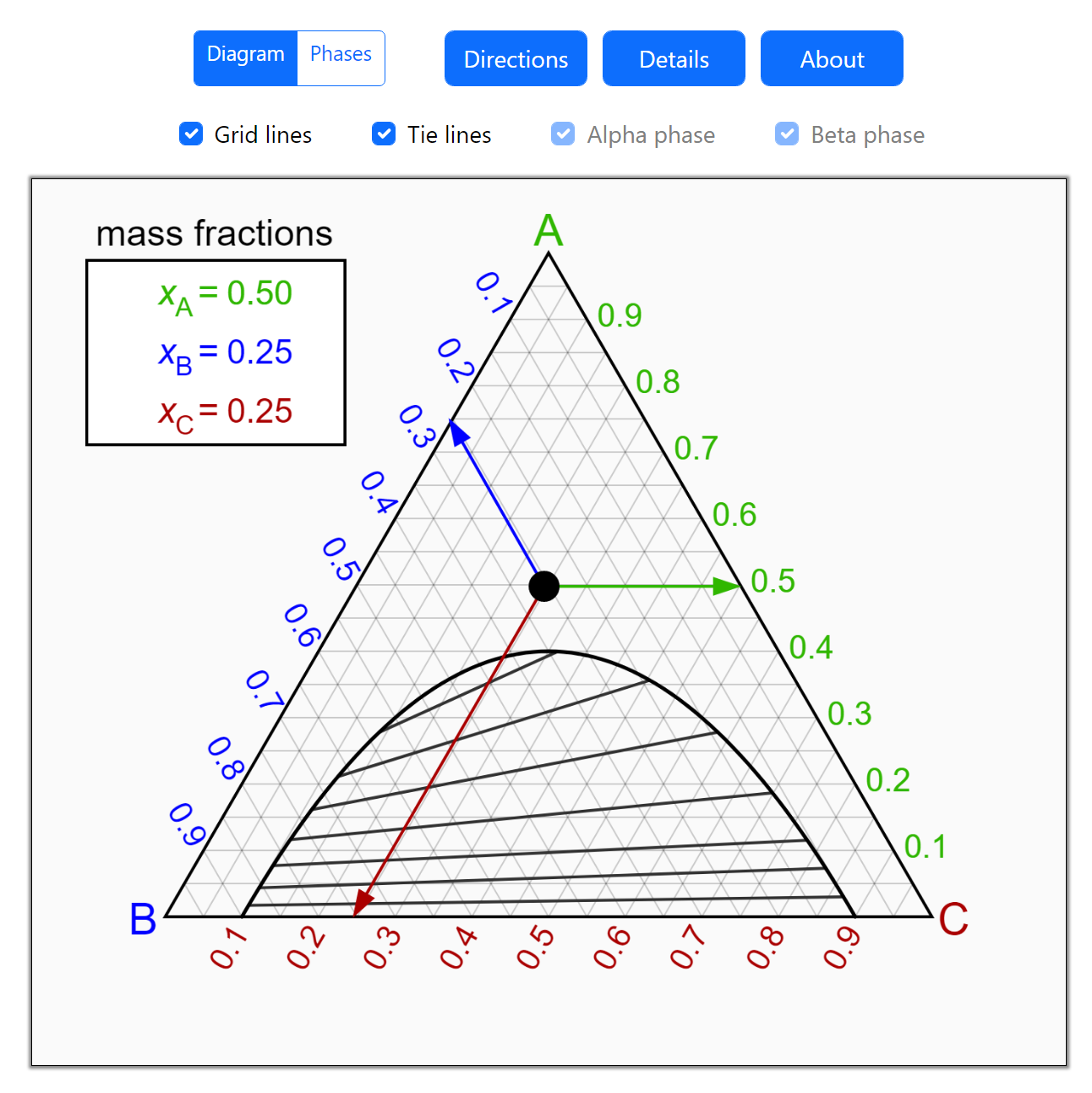
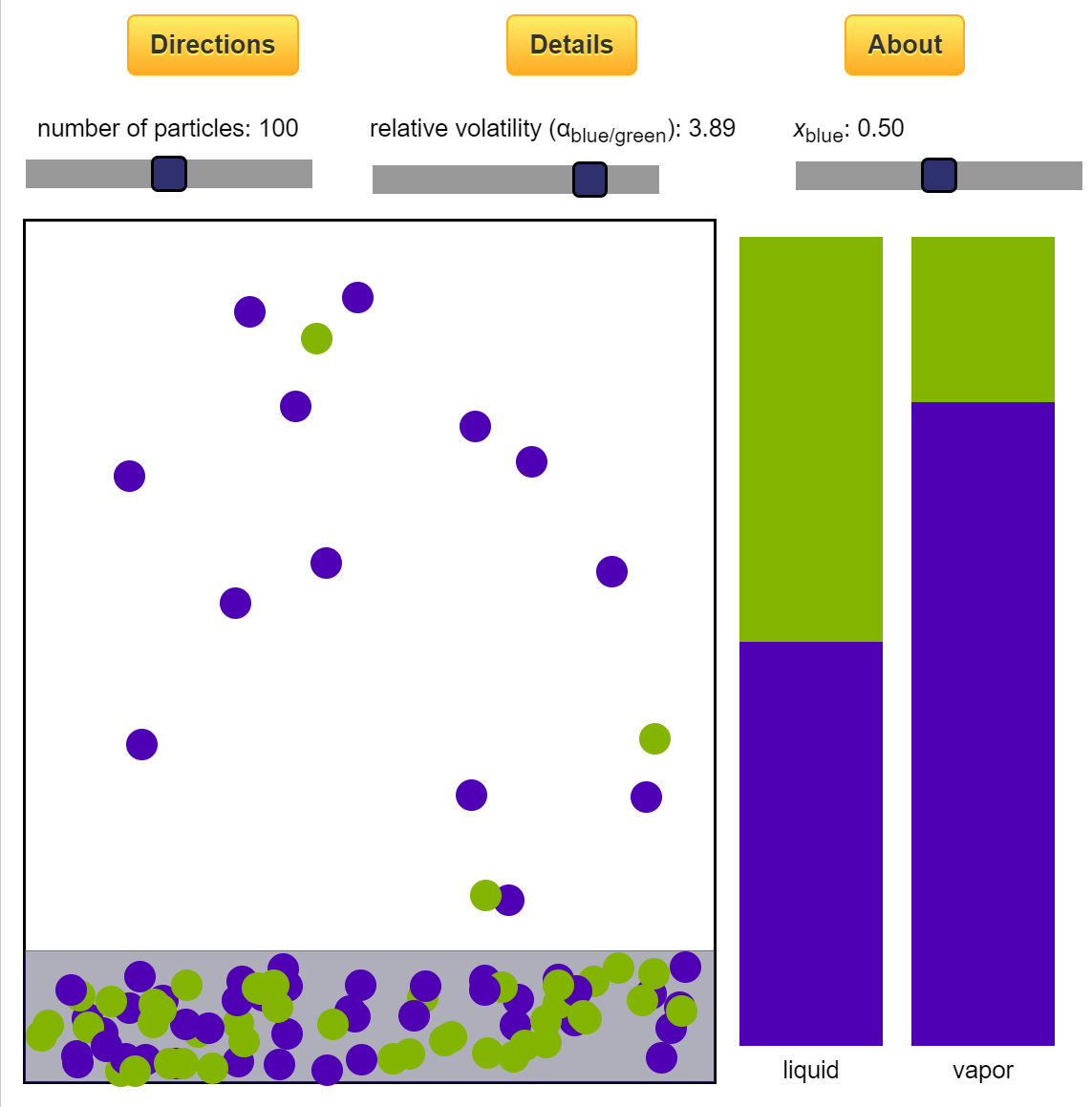
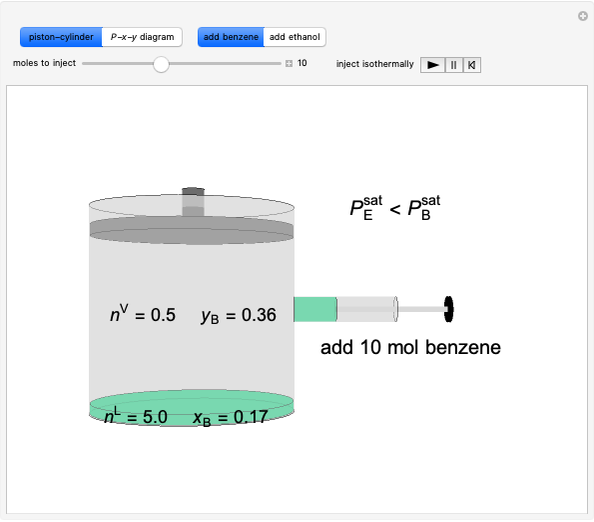
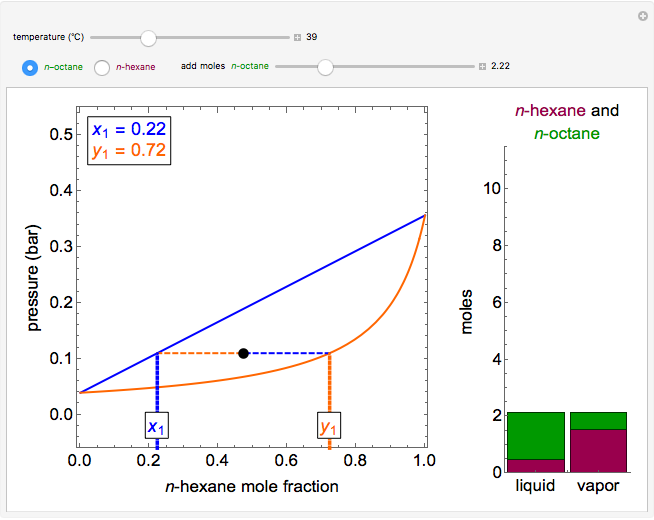
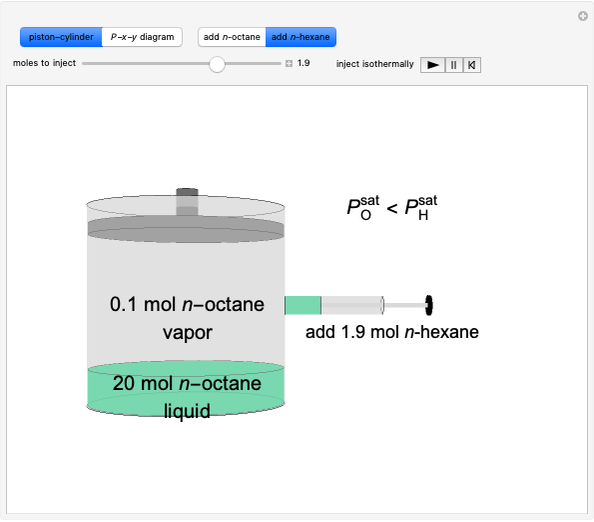
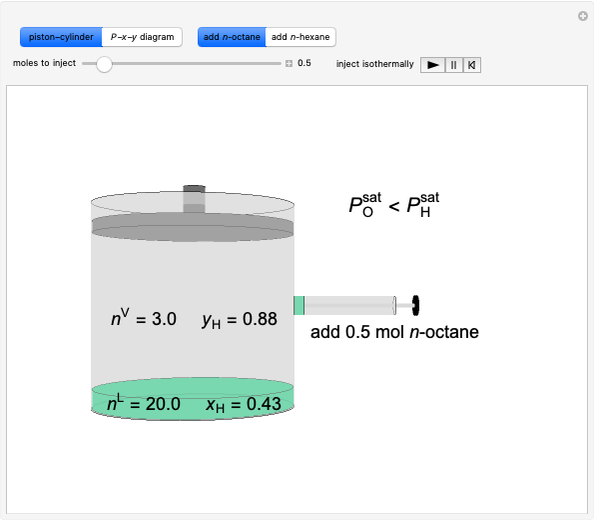
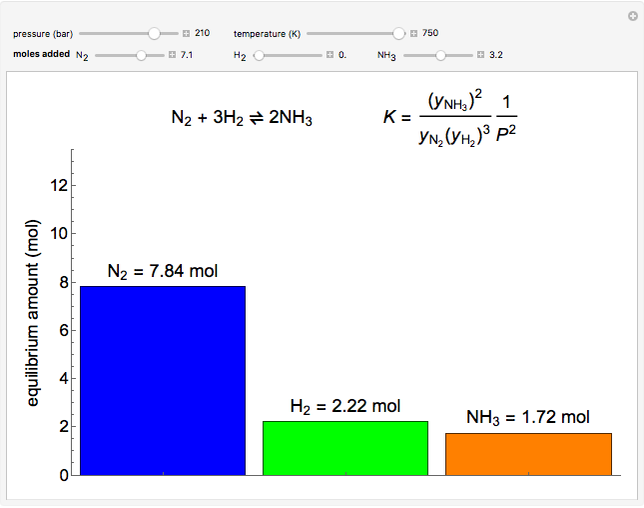
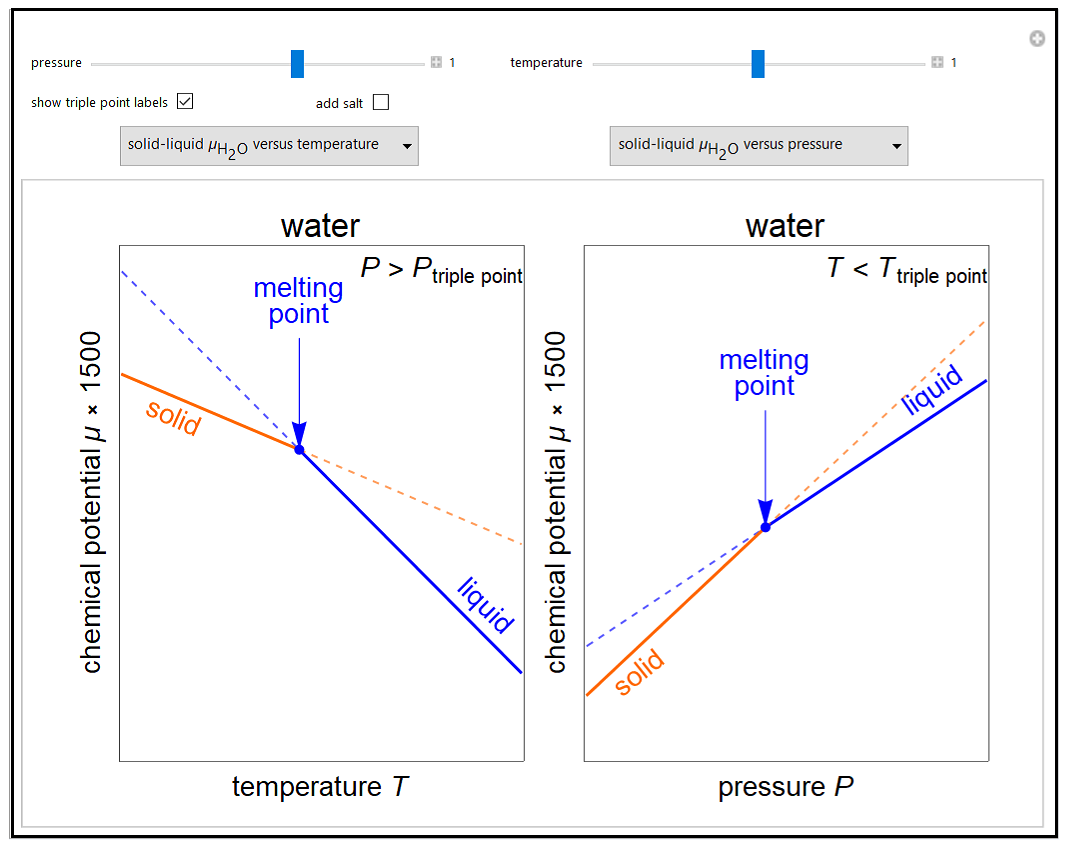
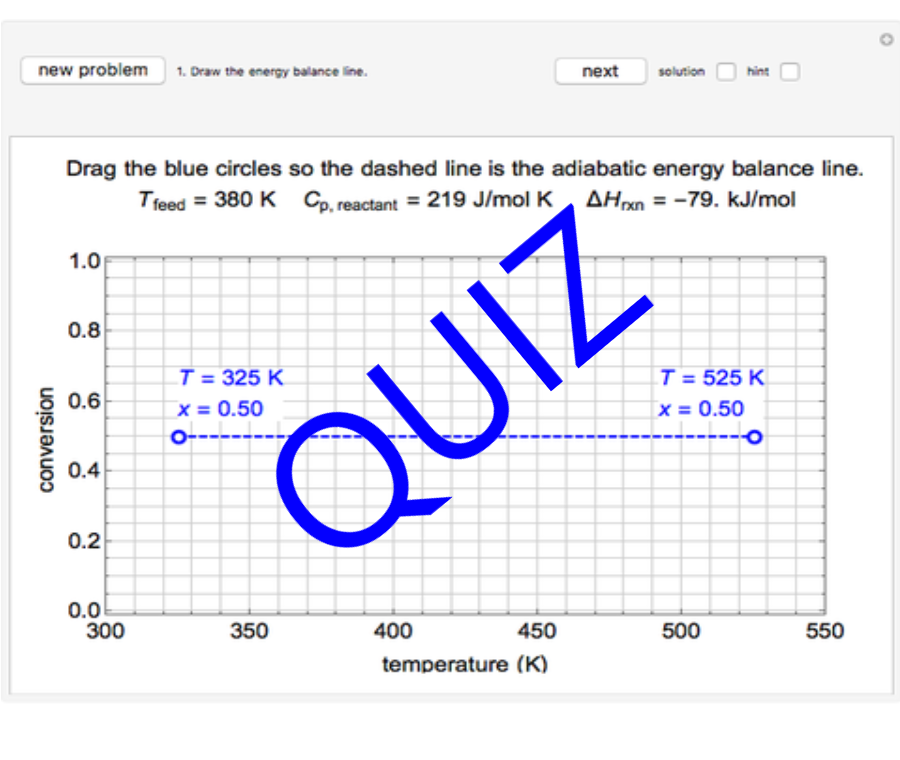
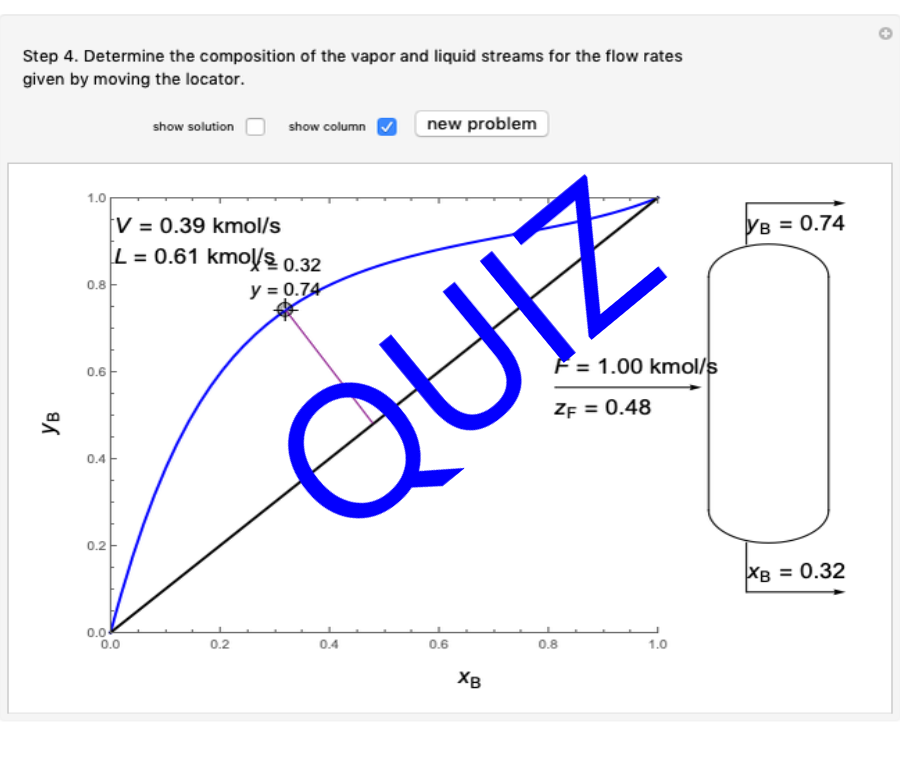
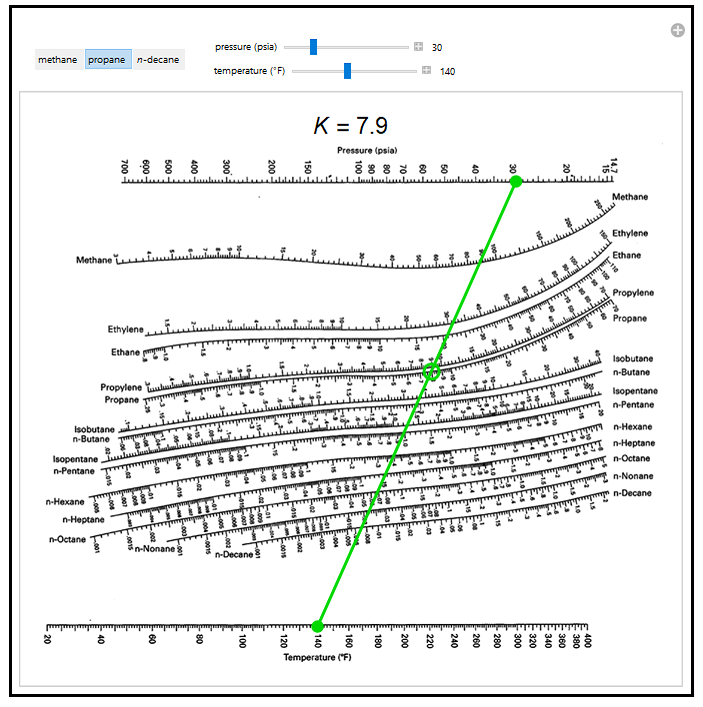
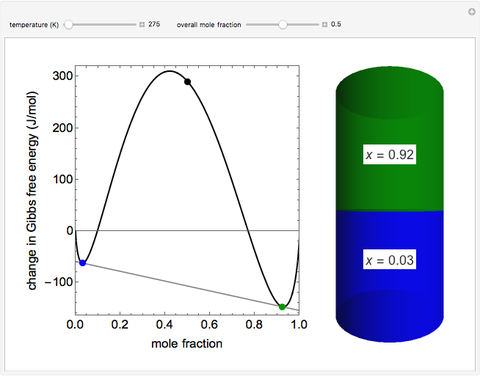
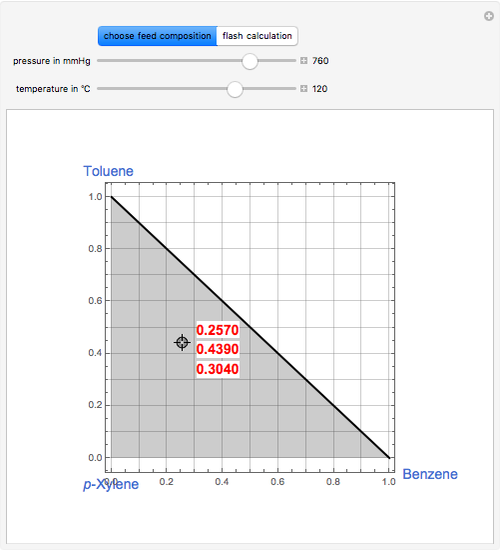
Thermodynamics 2 Simulations
- Select a category
- All simulations
- In-browser simulations
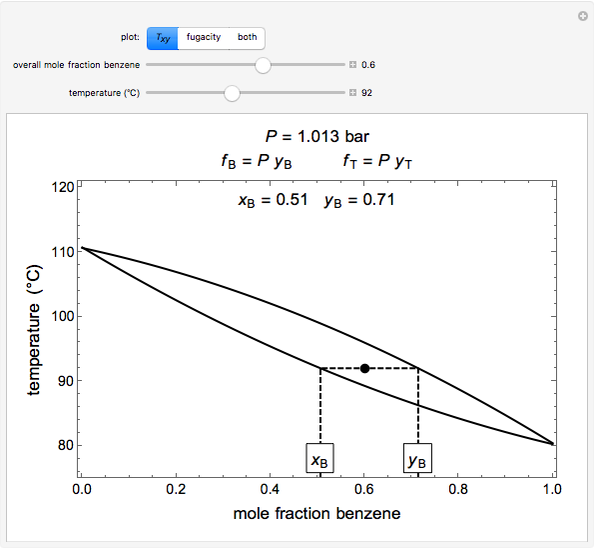
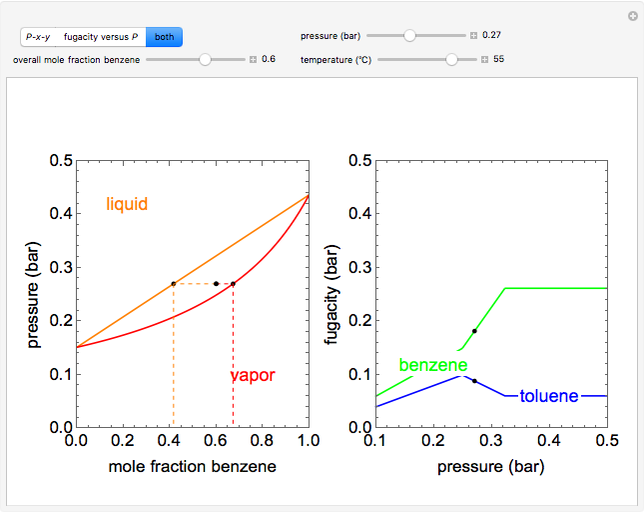
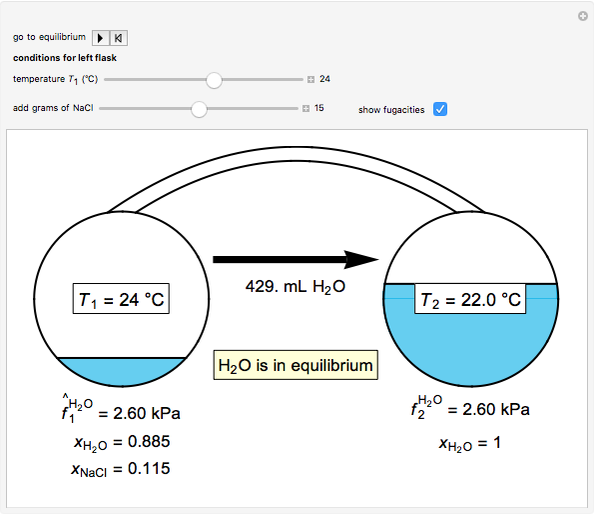
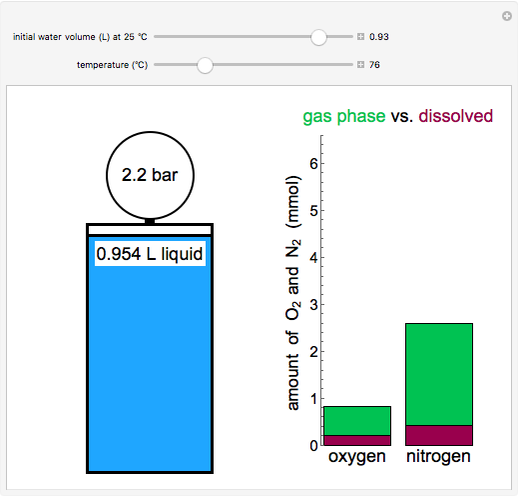
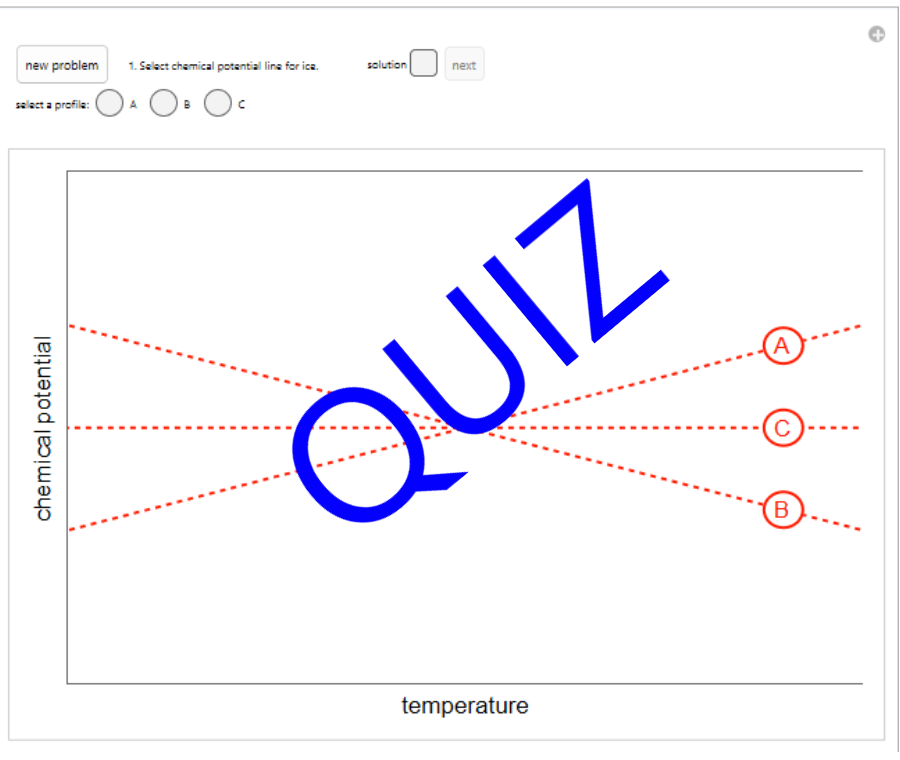
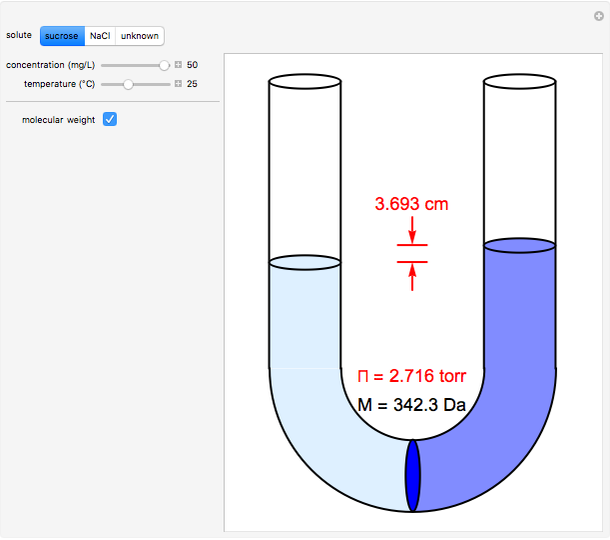
- Fugacity and chemical potential
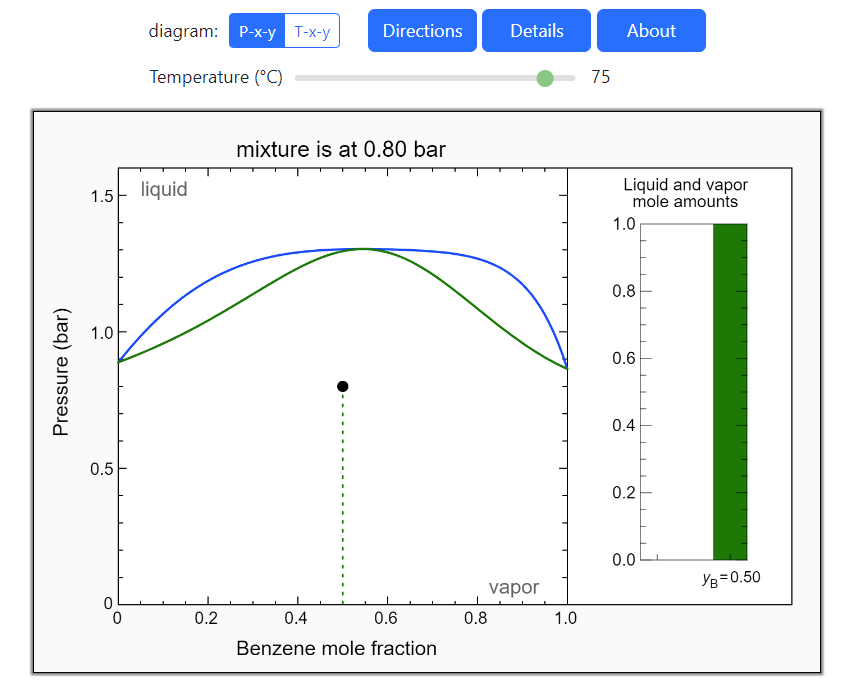
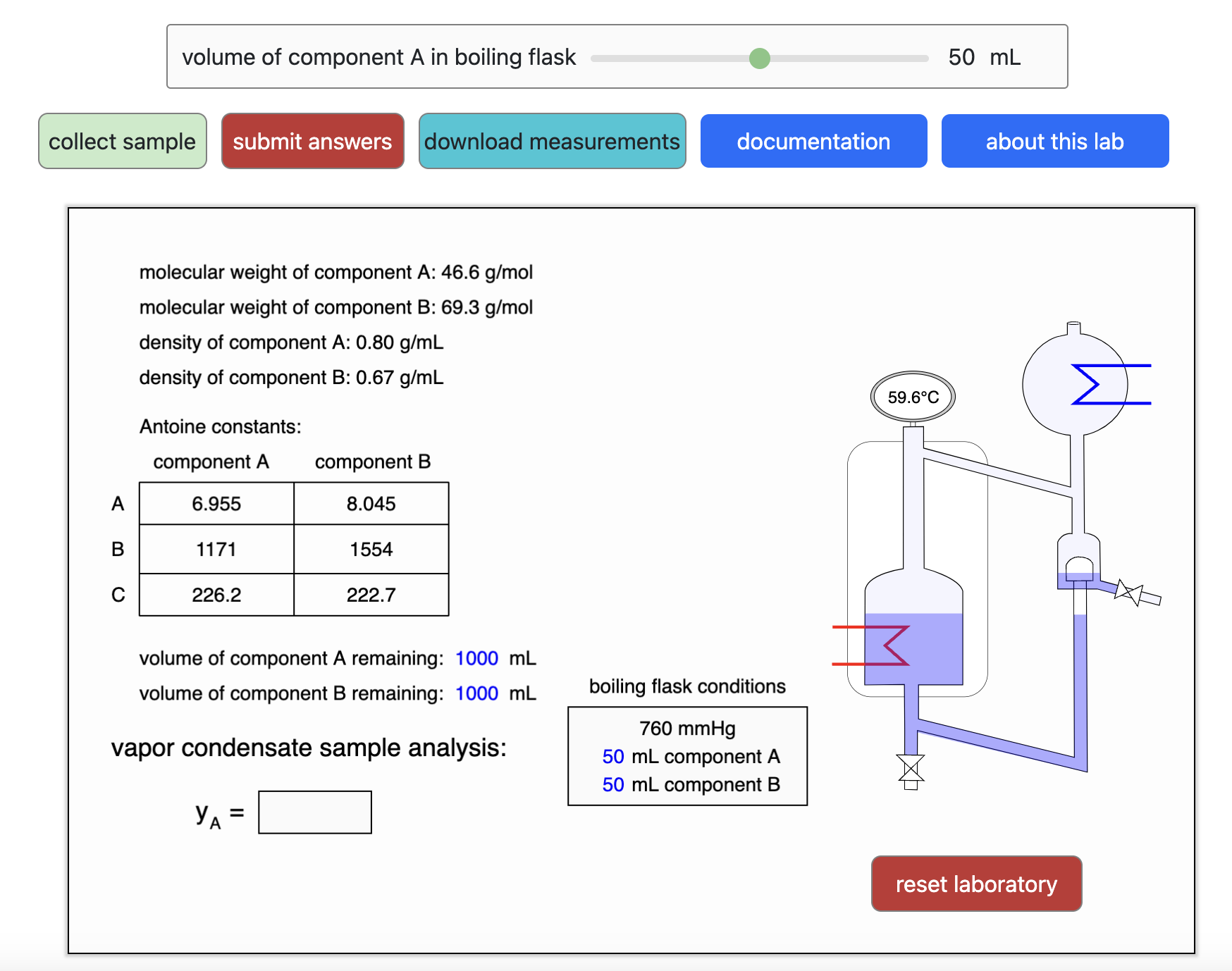
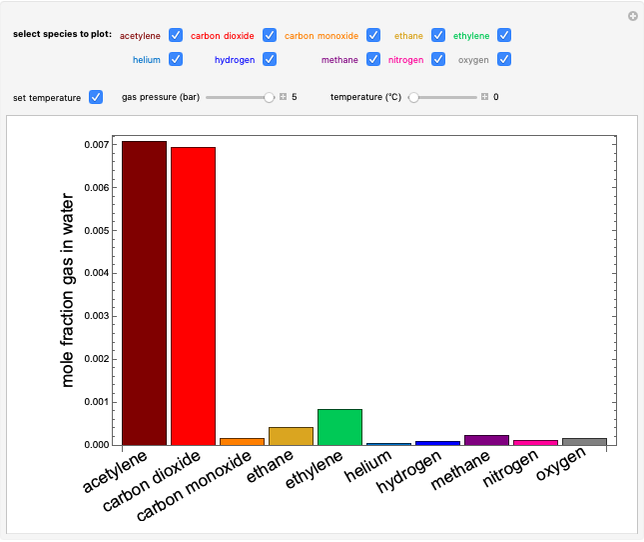
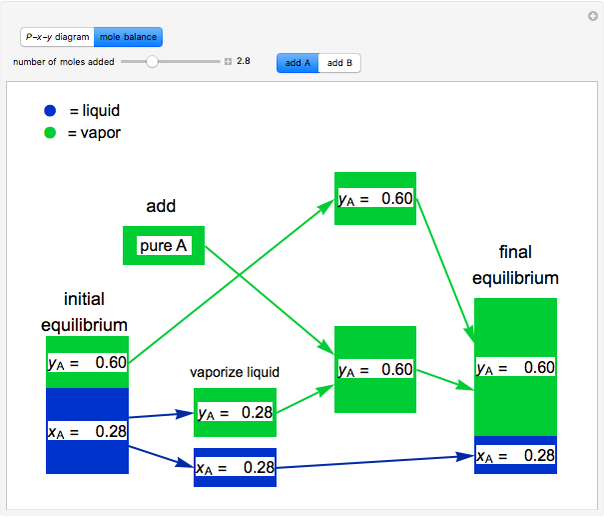
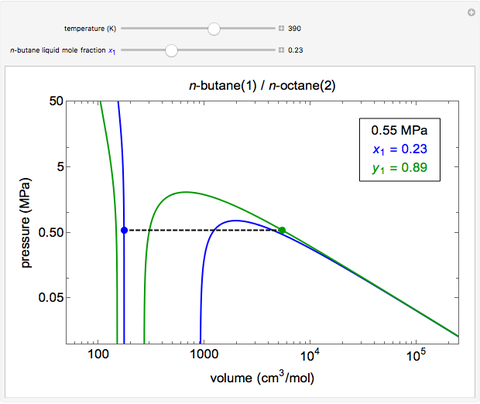
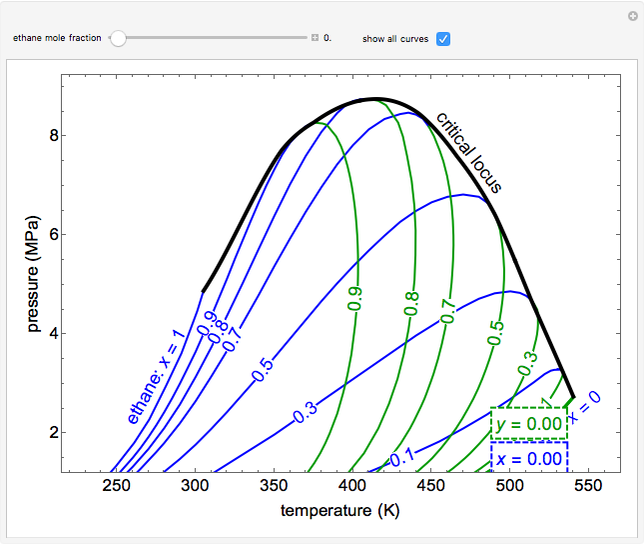
- Multi-component vle for ideal solutions
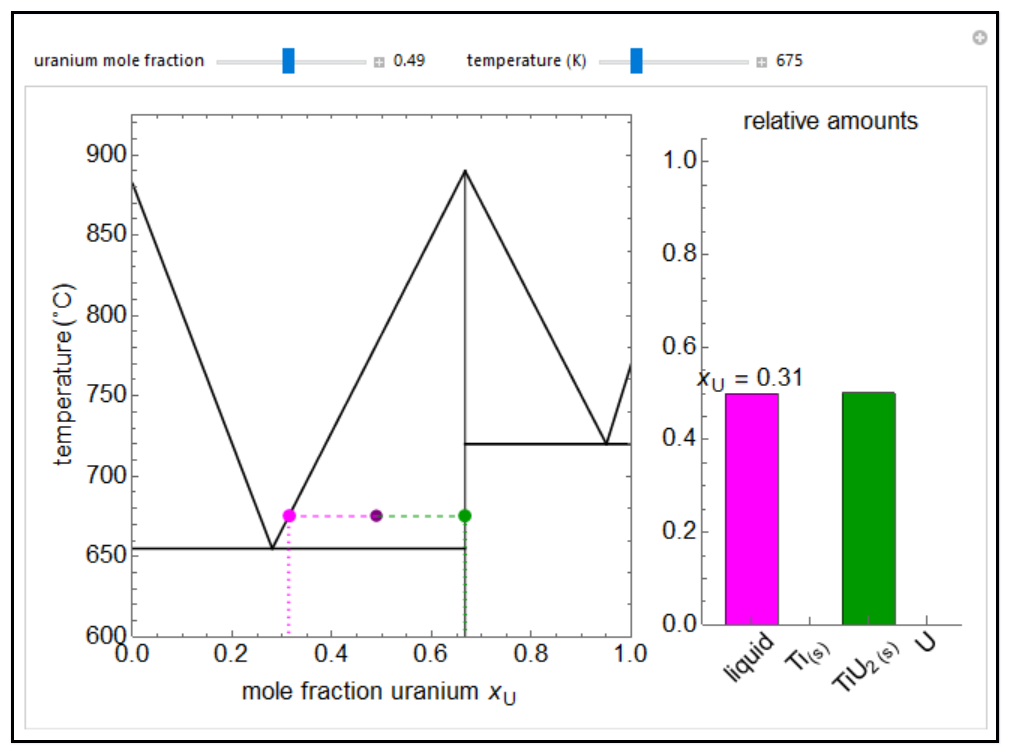
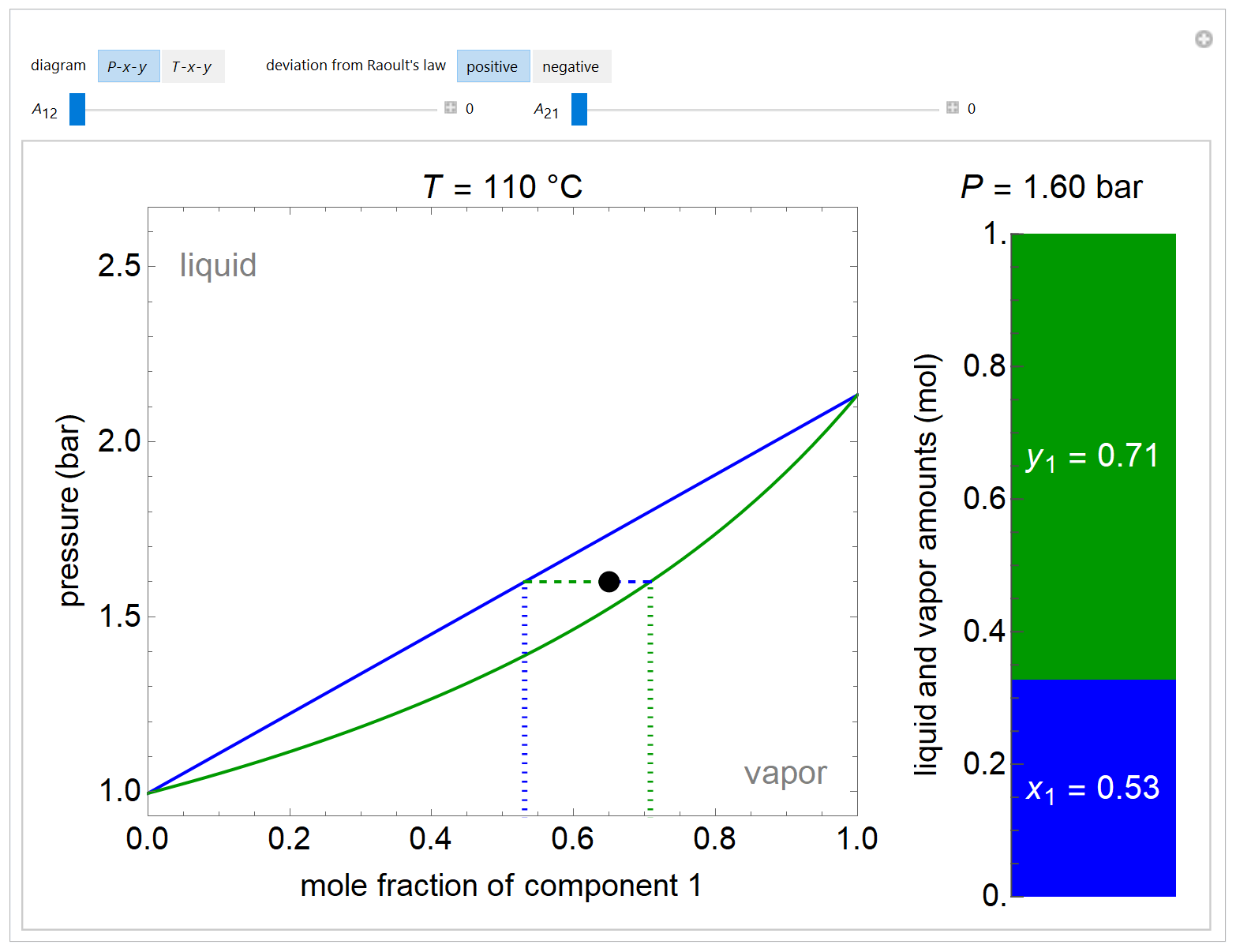
- Multi-component vle for non-ideal solutions
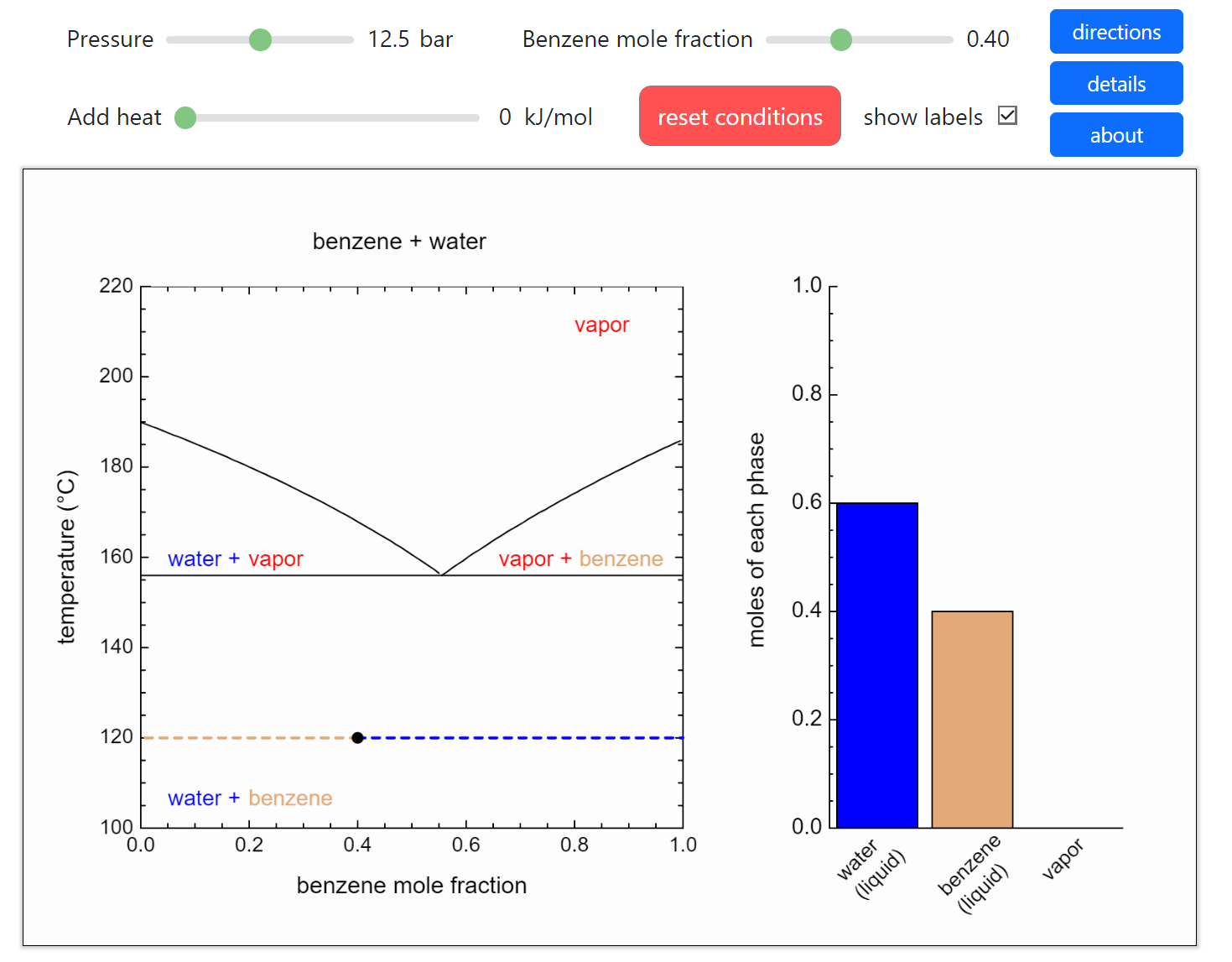
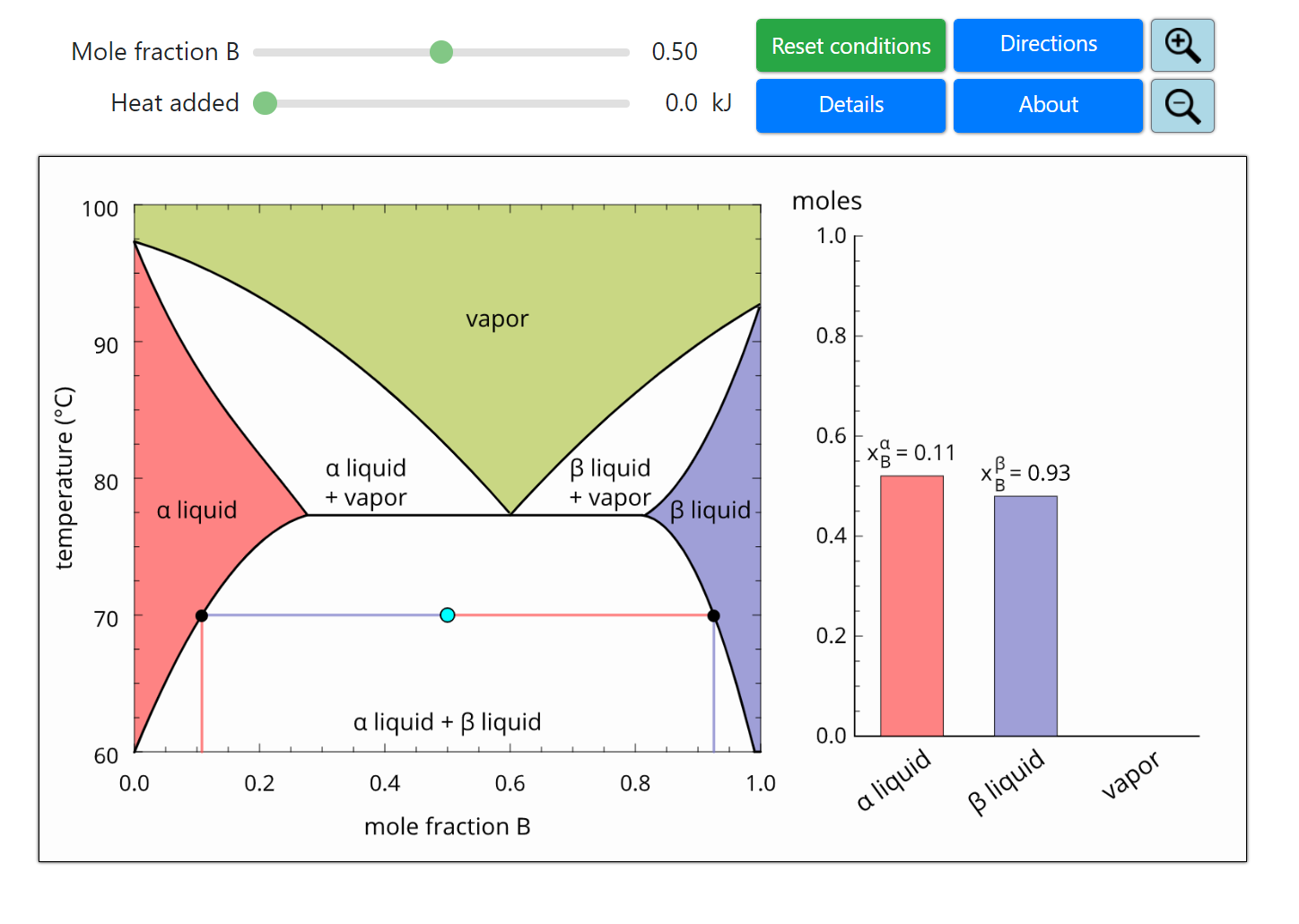
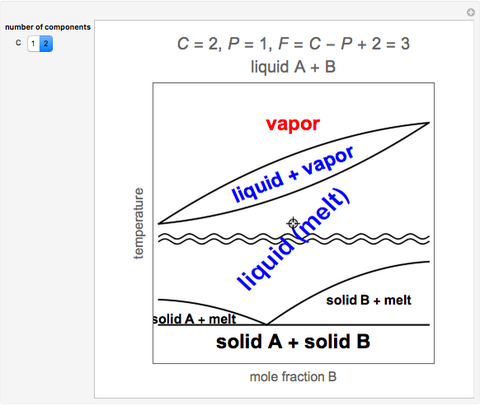
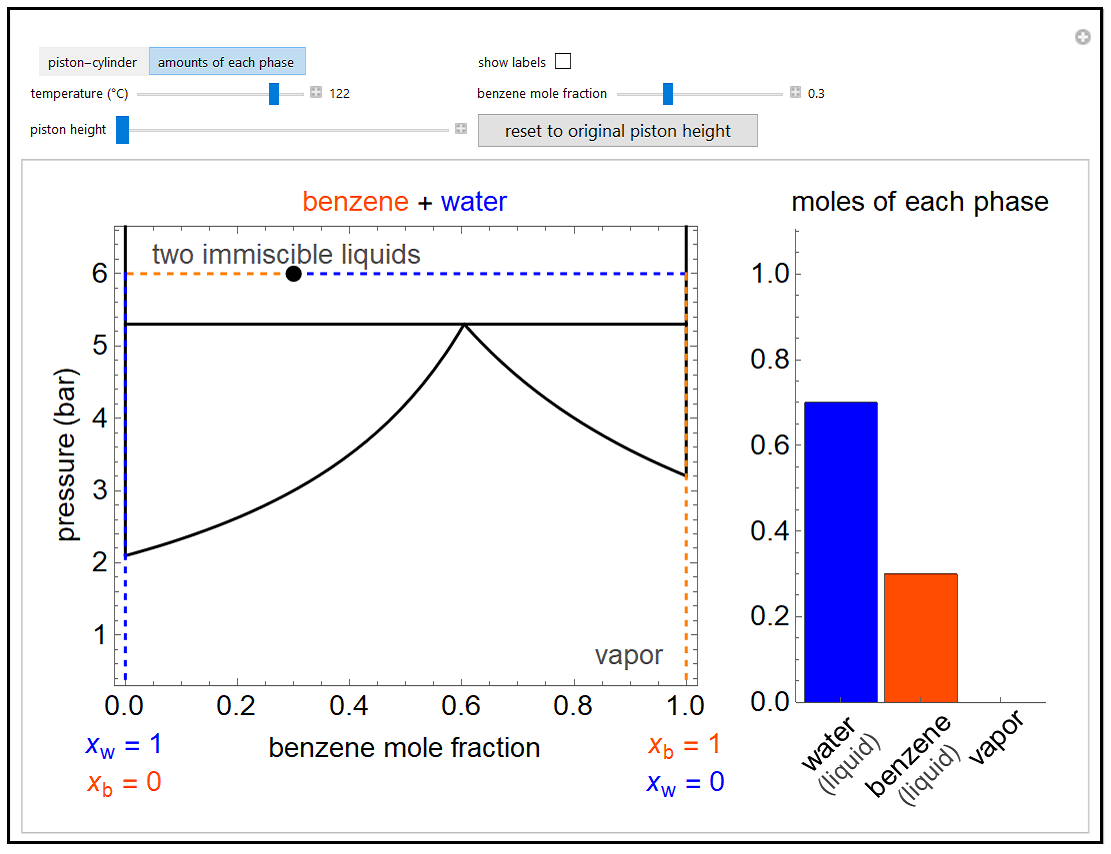
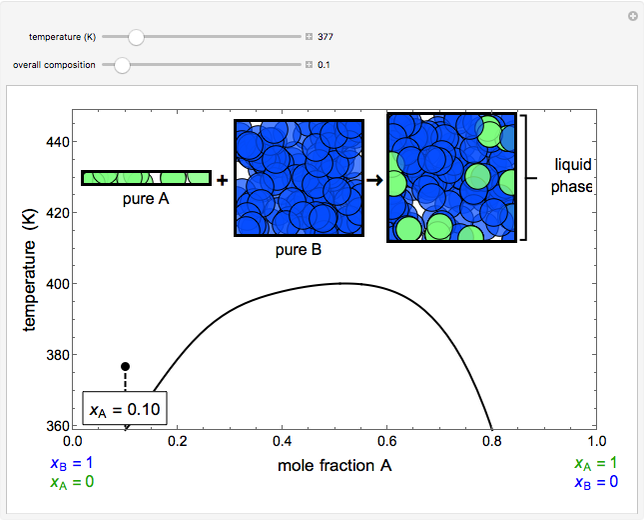
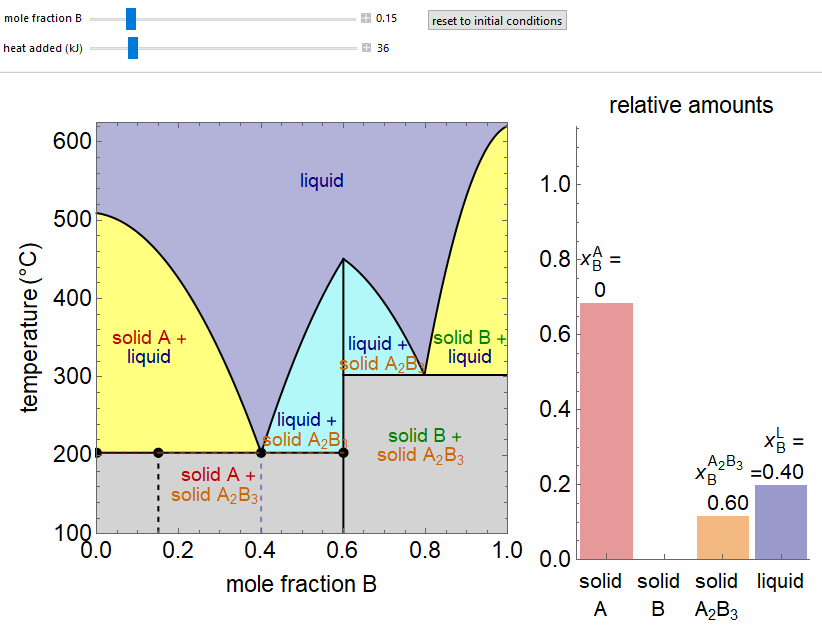
- Partially miscible and immiscible solutions
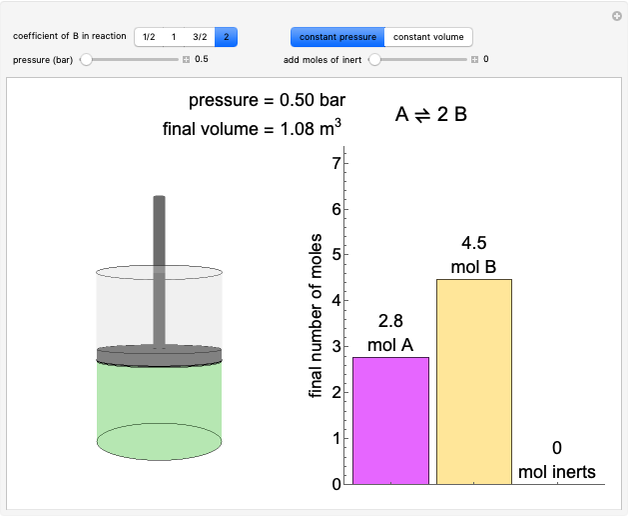
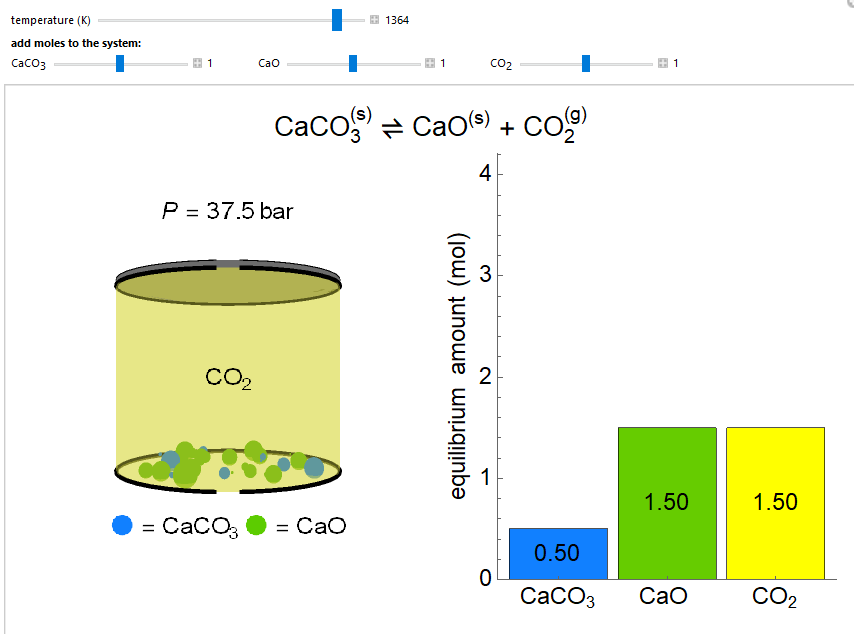
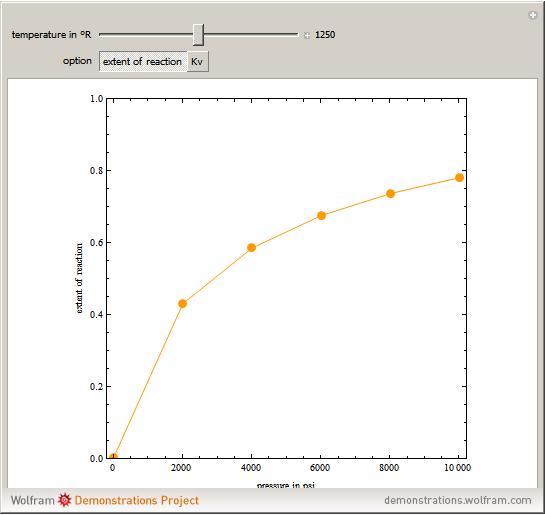
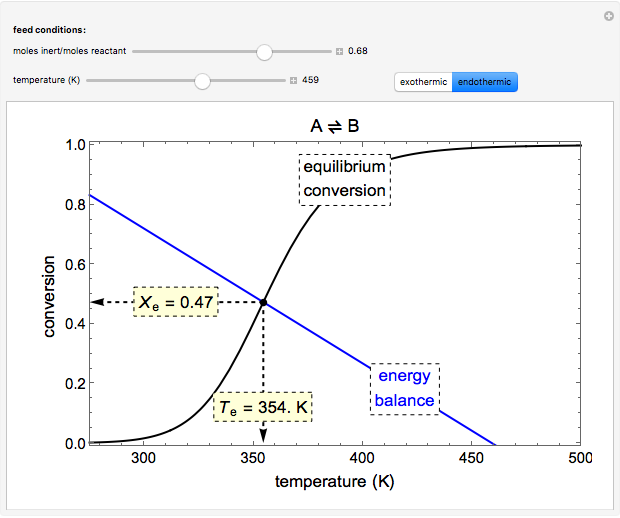
- Reaction equilibrium
No Items Found